Electronic Data Capture – Simple. Flexible. Proven.
secuTrial® is a professional, entirely browser-based GCP-compliant EDC system for collecting patient data in clinical or non-interventional studies and patient registries.
Since the year 2000, secuTrial® has been used in over 600 national and international studies and long-term projects conducted by university institutions, CROs, med-tech manufacturers and pharmaceutical companies – a clear testimony to its superior product quality, universal applications and high customer satisfaction.
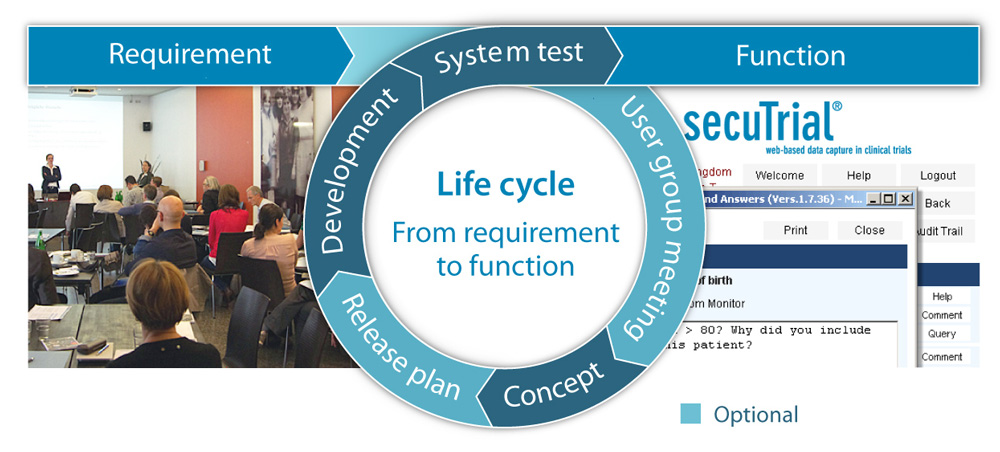
Customer requests are turned into secuTrial® functions as part of a validated process. For urgent projects, this can be done within just a few weeks outside of the regular release plan.
Key advantages of secuTrial®
- Range of applications
- Simple study setup
- User-friendly operation
- High quality standards
- Customer-oriented service
- Attractive price model
- Solutions proven in practice
- High investment security, future-proof solution