Job-Angebot
secuTrial®-User Group Meeting
 Our annual User Group Meetings: lectures, discussions, networking - and always in fine weather.
Our annual User Group Meetings: lectures, discussions, networking - and always in fine weather.
 More details
More detailsRegulations, validations & audits
GCP, 21 CRF Part11 + many other standards! secuTrial® is designed for licensing approval of pharmaceuticals.
 More details
More details
Key Data secuTrial®
Customer installations: > 50 Projects: > 660 Number of centres: > 3.000 Number of participants: > 14.500 Number of patients: > 250.000secuTrial® Flyer
Your Contact
secuTrial® patient apps – experience with electronic capturing of PRO
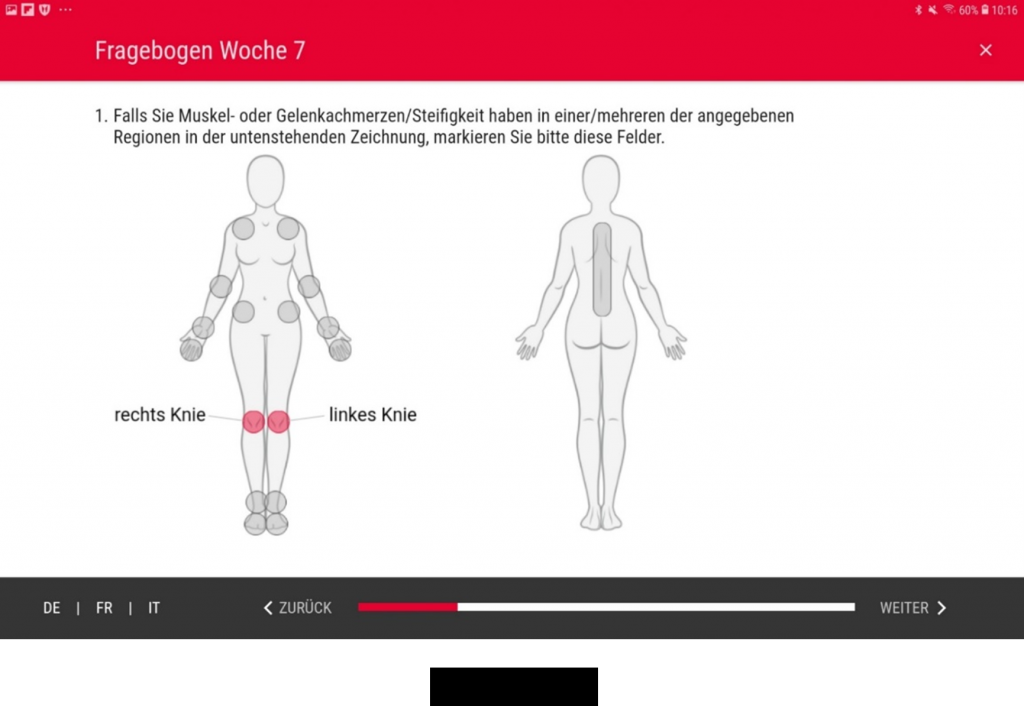
We recently developed four mobile secuTrial® patient apps for capturing patient-reported outcomes (PRO) in clinical studies. The design and user interface of each app were specifically tailored to the study and target group to ensure a smooth user experience and increase compliance. In terms of their use scenarios and required functions, the apps were very different. All of the apps are currently being used in studies.
For the Swiss Group for Clinical Cancer Research (SAKK) we developed a secuTrial® tablet app which has been in use since July 2019. The study is being conducted on clinic-owned tablets which are handed to the patients for documentation in the clinic during a total of 11 visits. The app was developed and optimised specifically for this (Android) tablet model.
By February 2020, the app had been used by 107 patients with breast cancer. The study subjects are aged between 35 and 80 years old. Where necessary, the study staff help patients with the process of logging in to the app. Even older patients have no difficulties filling in the detailed quality of life questionnaires. According to Mr Schulenburg from SAKK, they are able to complete them easily “thanks to intuitive operation and the excellent design”. Only on very rare occasions have technical problems occurred in transferring data to secuTrial® as a result of an unstable internet connection.
Although they were invited to use the app, about half of the participating study centres still opted for conventional paper-based capturing of patient data. Further testing and practical experience will no doubt help to establish electronic capturing over the course of time.
Another technically complex secuTrial® app was developed for CTC North (Hamburg) and its customer, the Clinic for Psychosomatic Medicine and Psychotherapy of the Medical Center Hamburg-Eppendorf. The app has been in use since November 2019 as part of a study on the early detection of depressive disorders, funded by the Innovation Committee of the Federal Joint Committee (G-BA). To date, the app has been used to recruit and screen over 6,000 subjects for depression in 50 GP surgeries. By July 2021, over 10,0000 people will be screened via the app.
The app was developed for offline use on (Android) tablets exclusively used for the study. Study centre staff in the GP surgery issue the tablets to patients after they have given their consent to participate in the study. The app users are a fairly heterogeneous group, ranging in age from 18 to 90. Thanks to the restrictive user guidance and large-scale layout, the app is easy to use even for older people. Technical questions and problems have only arisen on isolated occasions and were quickly resolved without requiring an app update.
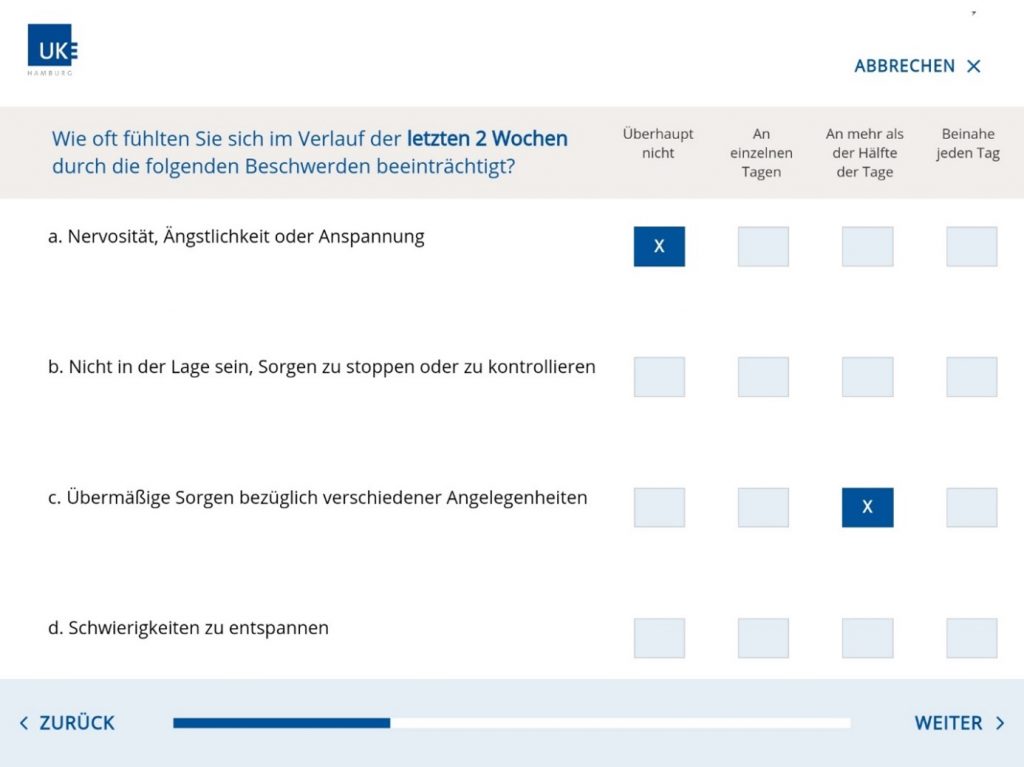
We also developed a secuTrial® smartphone app for the Gynaecology Clinic and Psychiatric Clinic of Universitätsklinikum Erlangen as part of a project funded by the Federal Ministry of Education and Research (BMBF). The app has been in use since March 2020 and is designed for capturing PRO in pregnant women. As the women use their own smartphones, we developed apps for iOS and Android which can be downloaded from the respective stores.
The women are recruited at the clinic. After giving their consent to participate in the study, they are given a brief introduction to the app and their login details. The patients then install the app independently at home and document a total of 31 visits over a period of 16 months. As an additional feature, the app offers mindfulness exercises that the women can do at any time and are part of the intervention. 107 women have been recruited so far (as at February 2021). The patients have been able to use the app without any issues and understand the structure and navigation. On the penultimate day of the 5-day data entry period for each visit, the users receive an entry reminder (local notification) on their smartphone. A small number of patients stated that they did not receive the reminders. A few patients also reported problems with logging in. However, after setting a new password, these issues were always able to be resolved.
As part of the TOTO study funded by the Federal Joint Committee, we developed a patient app for capturing patient-reported outcomes in cooperation with the University Medical Center Göttingen and Jena University Hospital. This was the first time that an app was based on the new secuTrial® Modern Data Capture, providing a highly comprehensive interface to secuTrial®. The app has been used reliably since November 2020 and has already been tested on multiple patients. Study participants are prompted by the app to document their sore throats weekly via push message.
We look forward to analysing the feedback from users.
We have also learned a lot during the development of the secuTrial® app and would like to share these insights with our customers even before we begin a new project together. We would therefore be pleased to advise you on the following points:
- Support: If patients are documenting independently at home, it is important to ensure adequate support (e.g. when patients forget their passwords, checking the settings for receiving notifications). As the app manufacturer, we also want to provide support because questions and errors inevitably arise when using apps which need to be addressed.
- Use of personal devices vs. use of uniform “study” devices:
Creating an app for just one specific “study” device is less expensive and involves fewer risks because the app only needs to be developed for one specific system (display size, operating system/version) and the study centre has control over all operating system updates (Android, iOS). If patients are using their own devices, the app must be compatible with as many (common) smartphones on the market as possible. This requires development for different platforms, observance of device/manufacturer-specific properties and extensive testing. Compatibility with future operating system versions (Android, iOS) is also essential.
- Updates: For longer study periods and use of patient-owned devices, app updates need to be planned because operating system updates (iOS, Android updates) and new devices inherently entail risks. When updating secuTrial®, compatibility with the individual apps must also be ensured.
Many of our clients see mobile apps as the future for capturing patient-reported outcomes. We’d be interested to know what you think. We look forward to hearing your views and will gladly answer any questions you may have.
Are you interested in finding out more or do you have any questions? Please feel free to get in touch with us.
Contact details interActive Systems
Posted in News
Leave a comment
Consolidating the source code of secuTrial®
For almost 20 years we have been developing and marketing secuTrial®.
Working together with our clients, we have implemented countless suggestions and ideas into a range of practical functions. The result is an extremely powerful and extensive software suite for clinical trials.
We are now looking ahead at the next 20 years and the many challenges resulting from the progressive development of technology. To ensure that our continually evolving software will effectively work in the decades to come, we inevitably need to enter into new territory.

Refresh icon vector.
For several years now we have been discussing strategies for consolidating the source code of secuTrial®.
In consultation with our management team, we have therefore decided to focus on systematically carrying out this consolidation in the coming release year 2018/2019. The development of new functions and the implementation of client requests will be postponed until the following release year. As well as creating a solid basis for future developments, we hope that these changes will help to simplify and streamline your daily work.
Posted in News
Leave a comment
secuTrial® donates first prize for report on TMF tutorial

Dr. Esther Schmidt (DKFZ) was picked as the winner of the prize draw for her report on participating in a TMF tutorial on the subject of data protection. The report was published in the tutorial description in the ToolPool health research portal. The second and third prizes went to Matthias Löbe for his report on the use of the ARX Data Anonymization Tool as part of his teaching work in Clinical Informatics at Leipzig University and Sandra Pasewald for her report on using MOSAIC products E-Pix, gICS and gPAS in the German National Cohort health study.
Prof. Dr. Rainer Röhrig (University of Oldenburg and member of the Board of Directors of TMF) and Prof. Dr. Ulrich Sax (University Medical Center Göttingen and spokesman of the TMF working group for IT infrastructure and quality management) picked the winners during a workshop presenting the health research of ToolPool on the occasion of the 62nd anniversary of the GMDS (German Association for Medical Informatics, Biometry and Epidemiology in Oldenburg). They were assisted by the independent prize draw helper Anna Dirks (a student of the University of Oldenburg and a member of the GMDS 2017 conference team).
The portal offers support with planning and setting up complex IT infrastructures in medical research. The aim of the competition was to encourage interaction and the exchange of experiences between the users of various tools and services. As an incentive, an Apple iPad, a Garmin smart watch and a Samsonite laptop rucksack were offered as prizes. The first prize was provided by secuTrial®, the other two prizes were supplied by Kairos and Clinical Guideline Services, whose products are also listed on ToolPool.
The ToolPool health research portal provides information on topics such as research data management, data protection, registries and cohorts, and mobile IT tools for medical research. It also brings together IT tools and services developed within the TMF or by other academic institutions and commercial providers. The content of the portal is based on the IT report of the TMF, which has been published annually since 2013.
The products are selected based on a catalogue of criteria devised by the TMF working group for IT infrastructure and quality management. Furthermore, the working group is responsible for checking that everything is up to date and for the strategic further development of the portal. The portal was developed by the TMF with the involvement of the users and is operated by the TMF.
For more information: www.toolpool-gesundheitsforschung.de
Posted in News
Leave a comment
secuTrial® supports Ärzte der Welt

Theoretically everyone in Germany should have health insurance.
In reality, however, a remarkably large number of Germans do not have health insurance and for many this has been the case for years. The reasons for this situation are as diverse as people’s individual stories. According to official statistics, approximately 80,000 men, women and children are currently living without health insurance in Germany, and estimates put this figure far higher. Furthermore, certain groups of people who are technically insured – for example many homeless people – often have difficulty accessing the healthcare system. Many of these people are chronically ill, have not been to a dentist in years or are suffering from mental problems.
For over a decade now, Ärzte der Welt e.V. (the German member organisation of Doctors of the World) has therefore been offering free medical care and consultations to people in Germany with limited or no access to the healthcare system.
It is not only German citizens who benefit from this initiative. The service is available to everyone, regardless of their residency status or country of origin. On request, the service can be used anonymously.
Right from the start of the open.med project, Ärzte der Welt has attached great importance to patient documentation and to collecting both medical and social data.
This data is extremely important in order to provide patients with appropriate treatment and also to supply political decision makers with reliable statistics. Since 2014, Ärzte der Welt has been using a local database which serves as an electronic patient file. A team of volunteers set up the system and helped to maintain the database.
Due to the growing requirements placed on the system resulting from mobile and inpatient care and multiple locations, the database began to reach its limits. After searching for a simple and stable centralised administrative solution, Ärzte der Welt found two partners willing to offer support: the clinical research organisation GKM and us. Since July this year, the patient file has been made available via secuTrial®. We are pleased to be able to support this social cause by providing and maintaining the new database system. Volunteer doctors and medical assistants can now access their patients’ data more quickly and easily from wherever they are working. By migrating to our professional system, the security of patient data has also been improved.
Posted in News
Leave a comment
500th trainee for secuTrial®

In September 2017 the 500th secuTrial® trainee completed their training at the Clinical Trials Center of the University Hospital Zurich. This was a very special milestone which we commemorated with a small gift.
The training modules offered by the CTC focus on creating eCRFs, data entry and validation, and data export. Each training session takes between 1.5 and 2 hours and an official training record is issued for the trial management file. Since 2010, a total of 390 training sessions have been organised with 1 to 5 participants.
Having worked with secuTrial® for many years, the CTC team at the University Hospital in Zurich have contributed to many of our innovations and further developments with their numerous ideas and suggestions. At our annual user meetings they have presented many interesting topics and are always very welcome as speakers.
We would like to express our special thanks to Dipl.-Ing. Dirk Smolinski and his team for their fantastic cooperation over the years and for kindly providing the photograph.
We wish all of the participants the best of success for their work with secuTrial®.
Posted in News
Leave a comment